Sleep Study Management
Personalized Clinician Study Management
Simplify and improve the sleep testing, diagnosis and treatment workflow for sleep clinicians and sleep labs with one solution for sleep study viewing, editing, and reporting.
Watch the video to discover the seamless workflow of EnsoSleep cloud-based sleep study management.
Experience Simplified Clinician Workflow
-
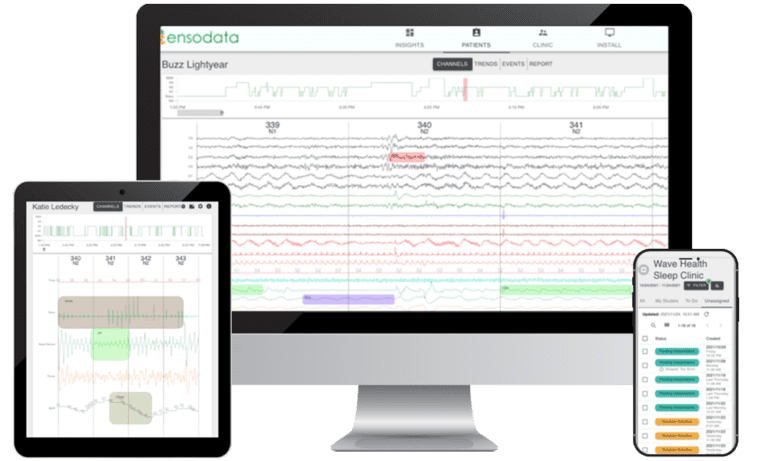
Seamless study review, editing, and reporting process accelerates the patient care journey
-
All studies available in one platform, regardless of software or devices
-
Automated emails to physicians include direct links to studies for review
-
Customize notification cadences to align with physician preferences
Manage All Sleep Studies in the Cloud
-
Sleep studies are centralized in one platform across all sleep lab locations
-
Remotely access sleep studies without a VPN on any computer, tablet, or device with a web browser
-
Securely access diagnostic data from all leading PSG and HSAT devices, regardless of the manufacturer
-
Eliminate downloading and uploading of studies
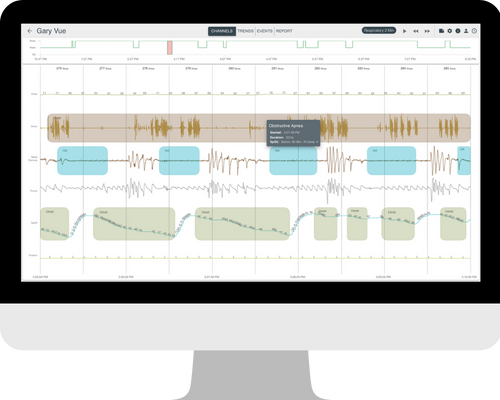

Discover Dynamic Reports for All Devices

-
One consistent report for physicians regardless of diagnostic device
-
Report text generation is automatic from the sleep study
-
Physician changes and edits are immediate and dynamic, pulling updated information into reports in real time
-
Actionable email reminders to physicians improve the patient care follow-up journey
Explore Personalized Clinician Study Management for Your Lab
Discover how EnsoSleep sleep study management provides clinicians with access to ALL studies and devices, across ALL locations, for ALL clinicians, in one interoperable, cloud-based platform.
Simply fill out our form and a team member will reach out to schedule a demo.



Request Demo
"*" indicates required fields
Free eBooks Highlighting the Impact of EnsoSleep Study Management
on Sleep Lab Owners, Directors, Managers, and Technologists
Learn why sleep lab owners and health system directors are turning to EnsoSleep Study Management to address core business priorities, including: increasing revenue, improving staff efficiency, and reducing scoring inconsistencies across the sleep lab.
Read why savvy sleep lab managers choose EnsoSleep Study Management to solve many common sleep lab challenges, including managing multiple devices, retaining staff, and finding ways to speed up the sleep test scoring process.
Discover how EnsoSleep Study Management helps sleep technologists overcome three growing sleep lab challenges: supporting increasingly complex patients, working with multiple devices, and taking on additional sleep lab responsibilities.
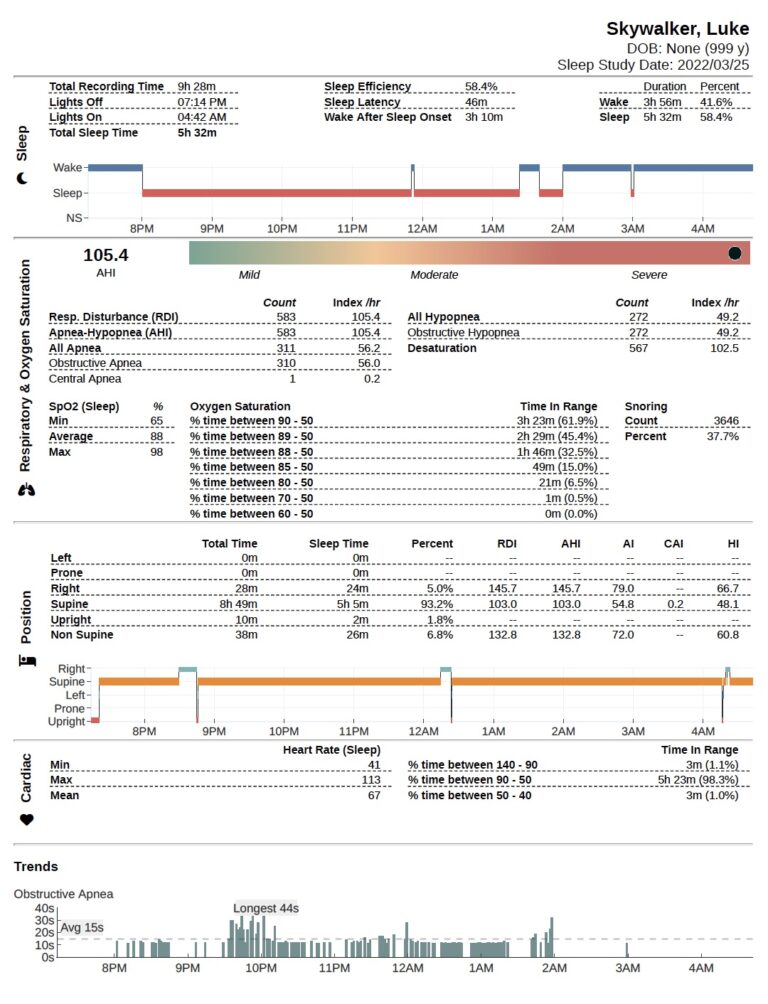
EnsoSleep FDA-cleared AI-assisted sleep scoring and analysis solution automates the scoring of studies, and reduces the time RPSGTs spend scoring PSGs and HSATs. Consistent, reliable results provide clinicians with more time to expand patient care, improve outcomes and treat more patients.
EnsoSleep produces an accurate Total Sleep Time (TST) for HSAT devices that do not calculate a measurement of sleep time. Including TST results in improved diagnostic accuracy for clinicians and patients and potential increased reimbursement.


Support the entire sleep lab team and improve the in-lab patient experience by providing scoring summary data just minutes after live sleep study recording. EnsoSleep Realtime Scoring optimizes the sleep lab workflow for split-night criteria, optimal titration pressure, or diagnostic on-the-fly scoring.

Accelerate the identification, diagnosis, and treatment of sleep-disordered breathing events with accurate, cost-effective, AI-driven analysis of photoplethysmogram (PPG) signals captured by pulse oximeters.




